How pH Influences Protein Separation in Ion Exchange Chromatography
Matthew Crawford
Ion exchange chromatography (IEX) is one of the most widely used techniques for protein analysis and purification. It exploits differences in the net surface charge of proteins, allowing separation based on subtle charge heterogeneities. Among the many factors that influence performance, pH is one of the most critical parameters because it directly alters both the charge state of the protein and the functional groups on the resin. In this post, we’ll explore how pH affects protein separation, and how these principles play out in both analytical-scale HPLC and purification-scale processes.
The Role of pH in Protein Charge
Proteins carry a net charge that depends on the ionization state of their amino acid side chains. The isoelectric point (pI) is the pH at which the protein has no net charge.
- At pH below the pI, a protein is positively charged and will bind to cation exchange resins.
- At pH above the pI, a protein is negatively charged and will bind to anion exchange resins.
By adjusting pH, we control both the strength of protein binding and the selectivity between species with different charge profiles.
pH Effects in Analytical-Scale HPLC
On the analytical scale, such as in IEX-HPLC for characterization, the pH has two main effects:
- Resolution of Charge Variants: Small changes in pH (as little as ±0.3 units) can alter retention times significantly. This is especially important for charge variant analysis of biotherapeutics, where acidic and basic isoforms may differ by only a fraction of a charge unit. A carefully controlled pH ensures reproducibility and sharp peak resolution.
- Peak Shape and Recovery: If the pH is too close to the protein’s pI, weak binding may lead to poor retention or broad/misshapen peaks. If it is too far from the pI, overly strong binding may require steep salt gradients for elution, potentially compromising resolution. Analytical HPLC can balance pH for both resolution and recovery.
pH Effects in Purification-Scale Processes
At preparative or manufacturing scale, the role of pH becomes broader than just resolution—it also impacts process economics, robustness, and scalability.
- Dynamic Binding Capacity: Proteins bind more strongly to the resin when their charge is well separated from the resin’s charge. Choosing a pH at least 1 unit away from the pI generally ensures good binding, maximizing resin utilization.
- Selectivity Between Proteins or Impurities: Adjusting pH can dramatically improve separation of target proteins from host-cell proteins, aggregates, or isoforms. This makes pH scouting an essential part of process development.
- Process Robustness: pH must be carefully controlled to avoid protein denaturation or aggregation. Many proteins are only stable in a narrow pH window, so purification buffers must balance optimal binding with product stability.
- Scalability Considerations: Unlike in HPLC, where short gradients and narrow columns dominate, purification-scale systems require large buffer volumes and longer equilibration times. The cost of buffers and resin lifetime is tied to the chosen pH, making it a critical process economics factor.

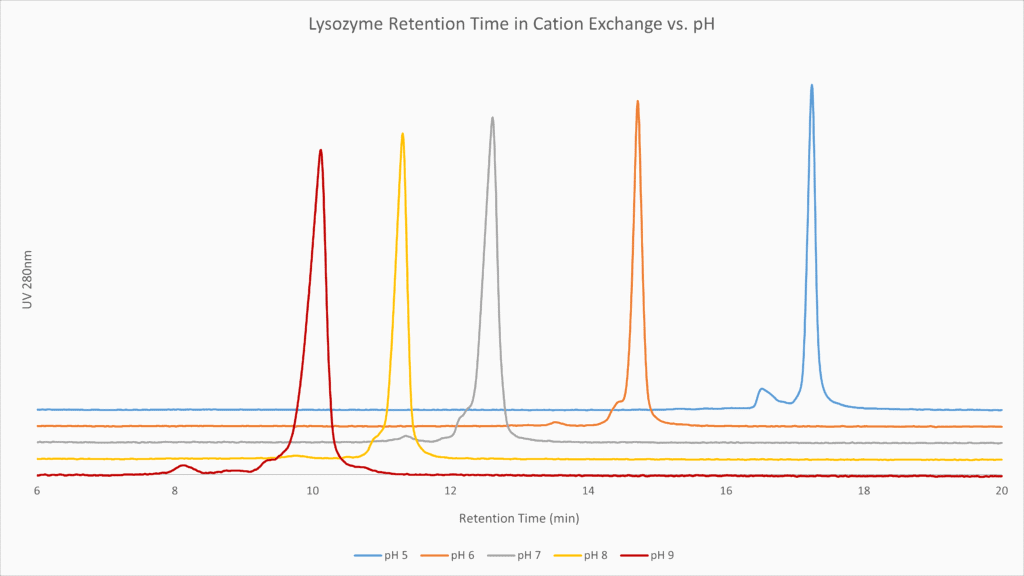
Figure 1: Lysozyme protein runs at 5 different pH’s over a strong cation exchanger from 0 to 1M NaCl. Note the differential resolution of contaminants but also the considerable improvement in peak shape at lower pH, due to the higher charge on the protein improving binding during loading.
Key Takeaways
- pH is one of two master variables in ion exchange chromatography (alongside salt concentration), controlling charge state, binding strength, and selectivity.
- In analytical HPLC, precise pH control ensures reproducibility and separation of subtle charge variants.
- In purification-scale IEX, pH influences binding capacity, impurity clearance, protein stability, and overall process economics.
Whether you are analyzing charge variants of a monoclonal antibody or scaling up a downstream purification process, careful optimization and control of pH is essential for successful ion exchange chromatography.
At Rilas, we leverage our experience in protein separations as well as high-throughput screening systems to generate the best methods possible for separation. As interest in protein-drug conjugates increases so too do the purification challenges associated with these advanced therapeutics. We purify the hard-to-purify across all modalities!
