EPSA Explained: What It Is, Why It Matters, and How It Accelerates Drug Discovery
Nathaniel Jorgenson
Polar surface area (PSA) is a widely used molecular descriptor in pharmaceutical research, closely linked to properties such as permeability, solubility, and oral absorption. In practice, PSA is most often estimated computationally from molecular structure, providing a convenient but simplified view of molecular polarity. However, calculated values do not always reflect how polarity is expressed experimentally, particularly for flexible molecules or those capable of intramolecular hydrogen bonding. As a result, there is growing interest in experimental approaches that can provide a more realistic assessment of polar surface area.
Experimental polar surface area (EPSA) is used as a practical measure of effective molecular polarity, helping to explain and predict behaviors that are not always captured by calculated descriptors alone. In drug discovery, EPSA is commonly applied to support permeability and absorption assessments, where subtle differences in exposed polarity can strongly influence membrane transport. It is also be used to rank and differentiate compounds with similar structures or calculated PSA values, identify the impact of intramolecular hydrogen bonding, and provide experimental context for structure-property relationship studies. By reflecting how polar functional groups are presented under experimental conditions, EPSA serves as a complementary tool in silico models, aiding compound prioritization and guiding optimization decisions throughout early discovery and lead development.
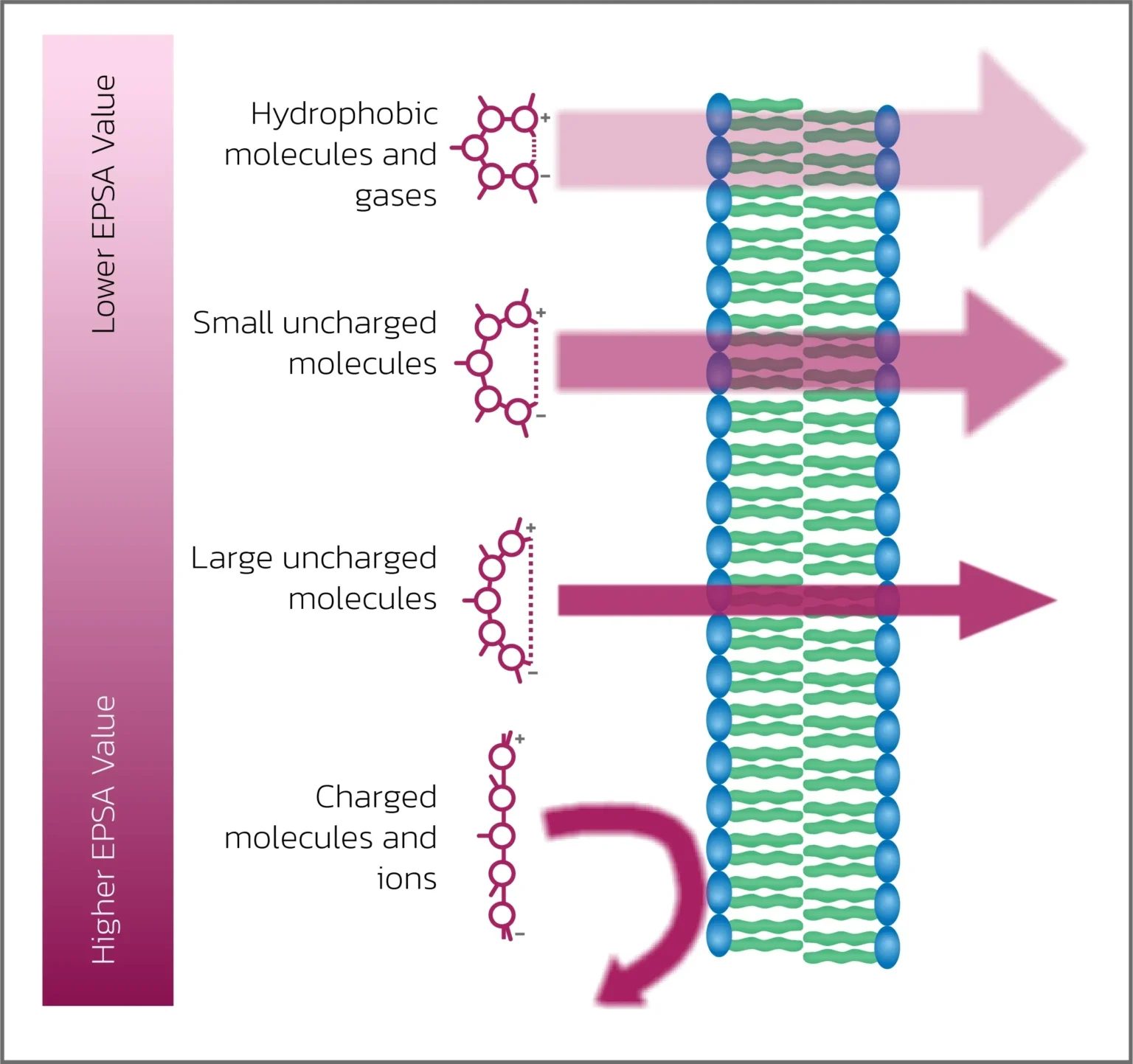
Figure 1: This schematic shows how EPSA relates to membrane permeability. Low-EPSA, hydrophobic or small uncharged molecules readily cross the lipid bilayer, while permeability decreases as EPSA increases. High-EPSA, charged molecules and ions interact poorly with the hydrophobic membrane core and show limited or no passive transport.
To experimentally assess molecular polarity, samples are analyzed using supercritical fluid chromatography (SFC) under standardized EPSA conditions. SFC provides a separation environment in which polarity-driven interactions between the analyte and stationary phase govern retention behavior. By running a set of well-characterized reference compounds alongside test samples, retention times can be directly linked to effective polar surface area. This approach enables rapid, reproducible assessment of polarity using chromatographic behavior rather than relying solely on calculated descriptors, forming the foundation for the separation shown below.
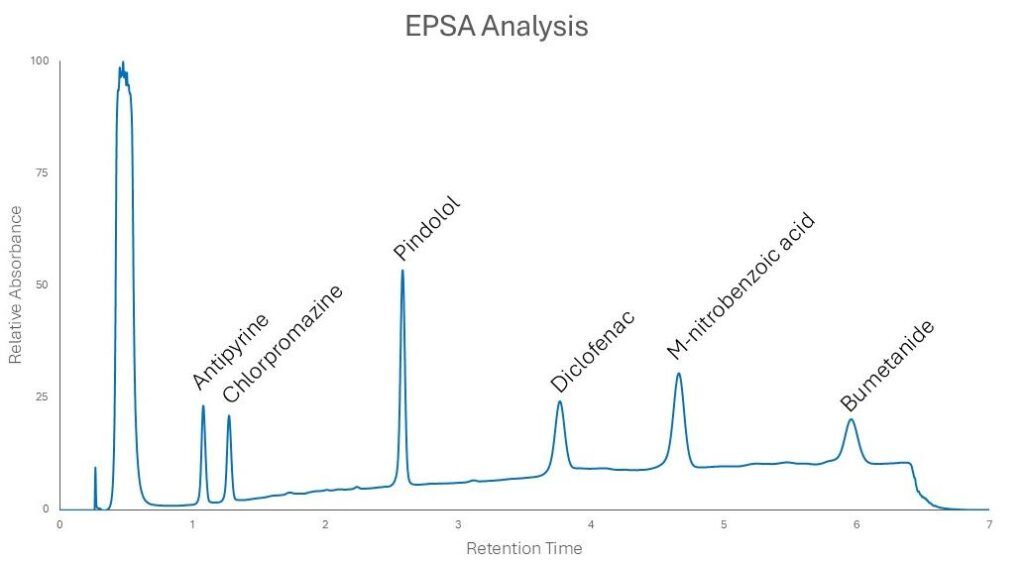
Compounds with lower EPSA values elute earlier in the run, reflecting weaker polar interactions with the stationary phase, while compounds with higher EPSA values are retained longer due to stronger polar interactions. Early eluting compounds such as antipyrine and chlorpromazine represent lower effective polarity, whereas later-eluting compounds like diclofenac, m-nitrobenzoic acid, and bumetanide exhibit progressively higher EPSA values. The clear ordering of retention times demonstrates how EPSA can be inferred directly from chromatographic behavior, providing a practical, experimentally driven measure of molecular polarity that complements calculated PSA values.
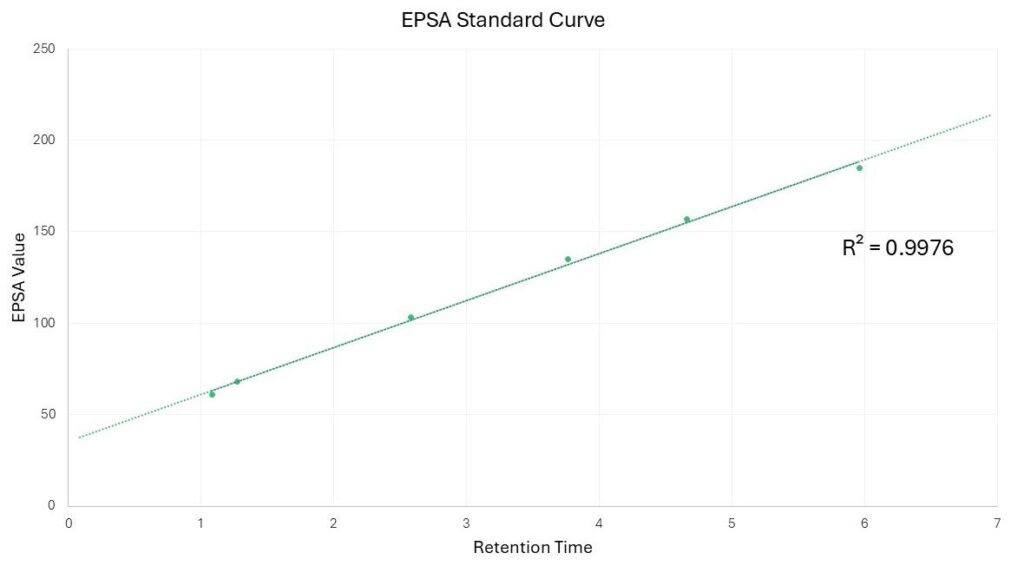
In summary, EPSA measurement by SFC offers a practical and experimentally grounded approach to understanding molecular polarity and its impact on key drug-like properties. By leveraging predictable retention behavior on an SFC system and anchoring measurements to well-defined reference standards, EPSA can be generated quickly, reproducibly, and in a format that is directly relevant to permeability and developability assessments. This makes SFC-based EPSA a valuable tool for supporting compound prioritization and structure-property relationships throughout early discovery and optimization efforts.
