The Impact of pH on Small Molecule Separations
Nathaniel Jorgenson
In liquid chromatography, mobile phase modifiers play a critical role in controlling analyte retention and peak shape. Mobile phase modifiers are small quantities of acids or bases added to chromatographic solvents to fine-tune the chemical environment of the separation. By altering parameters such as pH and ionic strength modifiers, directly influence retention time, peak shape, and reproducibility. Common examples include formic acid, trifluoroacetic acid (TFA), phosphoric acid, and acetic acid, each differing in acidity, volatility, and impact on analyte–stationary phase interactions.
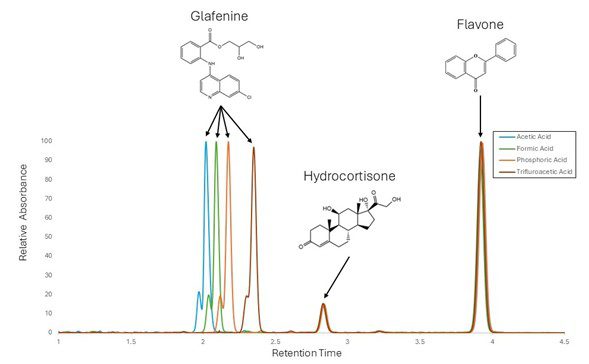
To explore the role of mobile phase modifiers on retention, we tested a three-component mixture containing glafenine, hydrocortisone, and flavone. Interestingly, while hydrocortisone and flavone showed stable retention times across all conditions, glafenine displayed measurable shifts depending on the modifier used. This contrast highlights how certain analytes are more sensitive to changes in mobile phase chemistry, making them valuable indicators of modifier effects on chromatographic behavior.

Figure 1: The figure shows an overlay of chromatograms for the three-component mixture of glafenine, hydrocortisone, and flavone. Each trace is plotted in a different color, corresponding to the use of 0.1% acetic acid (Blue), formic acid (Green), phosphoric acid (Orange), and Trifluoracetic acid (Red) as mobile phase modifiers. Hydrocortisone and flavone peaks align consistently across all conditions, indicating stable retention times, regardless of modifier used. In contrast, the glafenine peak shifts position between traces, with noticeable variation depending on the modifier used.
Among the three compounds analyzed, glafenine stands out because it contains a basic amine that can be protonated under acidic conditions. This protonation state depends on the acidic strength of the mobile phase modifier. When glafenine becomes protonated, its charge and polarity change, altering how strongly it interacts with the stationary phase and leading to shifts in retention time. In contrast, hydrocortisone and flavone lack protonatable amine groups and remain largely unaffected by small changes in mobile phase acidity. As a result, their retention times remain stable while glafenine’s shifts noticeably depending on which acid is used. The effect becomes even more pronounced when trifluoroacetic acid (TFA) is used, as the deprotonated TFA anion can form ion pairs with protonated glafenine, further increasing its hydrophobic character and causing a significant increase in retention time. It is also worth noting that TFA is the strongest acid of the 4 modifiers tested.  As a result, glafenine’s retention is highly sensitive to modifier choice, while the other two compounds remain stable across acidic conditions.
This behavior highlights why acidic modifier selection is important in small molecule chromatography. Even when acids are added at the same concentration, their strength and ability to form ion pairs can change how a compound interacts with the stationary phase. For compounds like glafenine that can be protonated, these differences can cause noticeable retention time shifts that must be monitored carefully when moving from batch-to-batch of mobile phase. Choosing the right modifier and keeping it consistent between runs helps ensure better reproducibility and method stability.
This impact of mobile phase modifiers is not only present in small molecule analysis. We have more information on how mobile phase modifiers impact protein purification in the blog post: How pH Influences Protein Separation in Ion Exchange Chromatography.Â
