Why Protein A HPLC-UV Should Be in Every Biologics Developer’s Toolbox
A CRO’s perspective on faster, more informative antibody analytics and processes…
Matthew Crawford
As a CRO supporting biologics developers from early discovery through late-stage manufacturing, we’re constantly asked the question: How can we get reliable antibody data faster, with less sample prep and more confidence?
One of the most common answers we give is protein A chromatography with UV detection. While Protein A chromatography is best known as a capture step in large-scale antibody purification, its analytical implementation (especially at the micro-scale) has become a powerful tool for decision-making across biopharma and biotech programs.
In this post, I’ll explain why we offer Protein A HPLC-UV as a service (in addition to our standard protein A FPLC purification services), how it differs from non-affinity chromatography/HPLC, and why it’s uniquely suited for crude samples in early discovery, rapid turnaround at high throughput in process development, and high-specificity mAb measurements across all levels of mAb drug development.
What Is Protein A HPLC-UV?
Protein A HPLC-UV combines: Affinity chromatography using an immobilized, engineered Protein A ligand, high-performance liquid chromatography (HPLC) for reproducibility and resolution, and UV detection (typically at 280 nm) for direct quantification of antibodies. Protein A binds specifically to the Fc region of IgG antibodies (and many Fc-containing formats), allowing selective capture directly from complex matrices such as cell culture supernatant, lysate, or partially purified intermediates. At the analytical scale, this means you can inject microliters of crude samples and obtain clean, quantitative antibody data in minutes, not hours or days.
How Protein A Chromatography Works (and Why It’s Different in HPLC)
Affinity vs. Non-Affinity Chromatography
Most chromatographic methods such as ion exchange (IEX), size exclusion (SEC), reversed-phase (RP) or hydrophobic interaction (HIC) separate molecules based on general physicochemical properties, such as charge, size, and hydrophobicity. These approaches are powerful, but they rely on differences between species to achieve separation. In crude samples, overlapping properties often lead to co-elution of host cell proteins and other contaminants, require extensive sample preparation to prevent competitive binding, reduce specificity and reproducibility and longer method development and method run times.
Protein A chromatography is fundamentally different.
Instead of separating based on bulk properties, Protein A uses biological recognition. Protein A ligand specifically binds the Fc region of antibodies while non-antibody components largely flow through. Antibodies are then selectively eluted under controlled conditions (e.g., low pH).
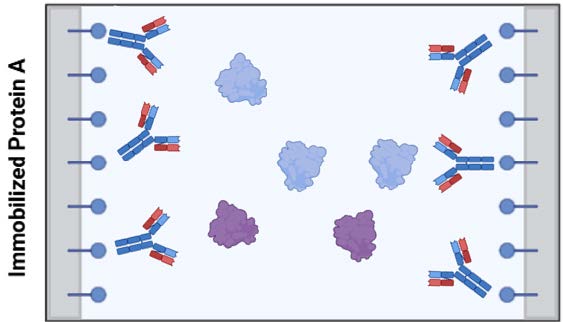
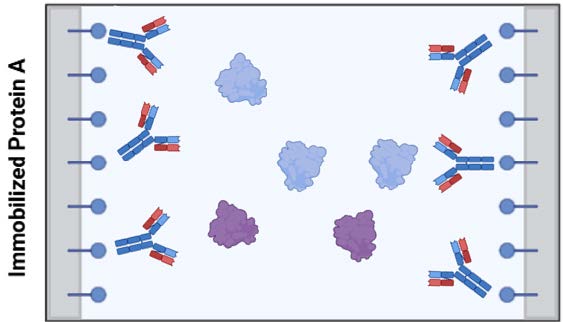
Figure 1: Immobilized protein A on solid support specifically binds IgG-like proteins, while letting other proteins (here represented as blue and violet generic globular proteins) flow through to waste. Bound proteins elute when pH drop changes charge-charge and shape interactions between Fc region and protein A.
The result is a process with exceptional specificity, high tolerance for crude samples, while requiring minimal (or no) sample prep and yielding consistent, platform-ready methods with no modifications required. For CRO workflows supporting multiple clients and molecule formats, that platform nature is invaluable.
Why Protein A HPLC-UV Excels with Crude Samples
From a CRO perspective, one of the biggest advantages of Protein A HPLC-UV is its ability to handle real-world samples, not idealized ones. We routinely analyze clarified or unclarified cell culture supernatants, early upstream process samples, raw lysates from expression screening, and process development fractions. All of these real-world samples contain large amounts of contaminants, often with very low concentrations of target analyte. Because Protein A selectively captures antibodies, molecules like host cell proteins, DNA, media components, and metabolites largely fl ow through. This means that during elution, the UV signal corresponds predominantly to antibody content (not other proteins) leading to robust quantitation even when total protein background is high. This is where non-affinity methods often struggle or require extensive cleanup steps that slow programs down.
Core Applications We Support
1.Micro-Scale Antibody Purification
Protein A HPLC enables micro-scale purifi cation (μg to low-mg range) directly on an analytical system. This is ideal for early discovery and screening where time is critical, there is often limited material availability, and the need for very rapid generation of purified antibodies for downstream assays is pressing.
Compared to gravity columns or spin fi lters, HPLC-based Protein A purifi cation offers: higher reproducibility with better recovery tracking (less reliant on scientist reproducibility). It can also provide better integrated analytics (UV, peak shape, retention time) for AI or other data-driven programs, which further benefits from its faster turnaround at high sample throughput when compared to gravity or spin systems.
2.Antibody Titer Measurement
Protein A HPLC-UV is widely used for accurate antibody titer determination in cell culture supernatants. It has several distinct advantages over methods like UV-Vis spectrophotometry, such as direct measurement without ELISA development, high specifi city for Fc-containing molecules, a wide dynamic range (less need for time consuming dilution series), and excellent method transferability across IgG-like molecules.
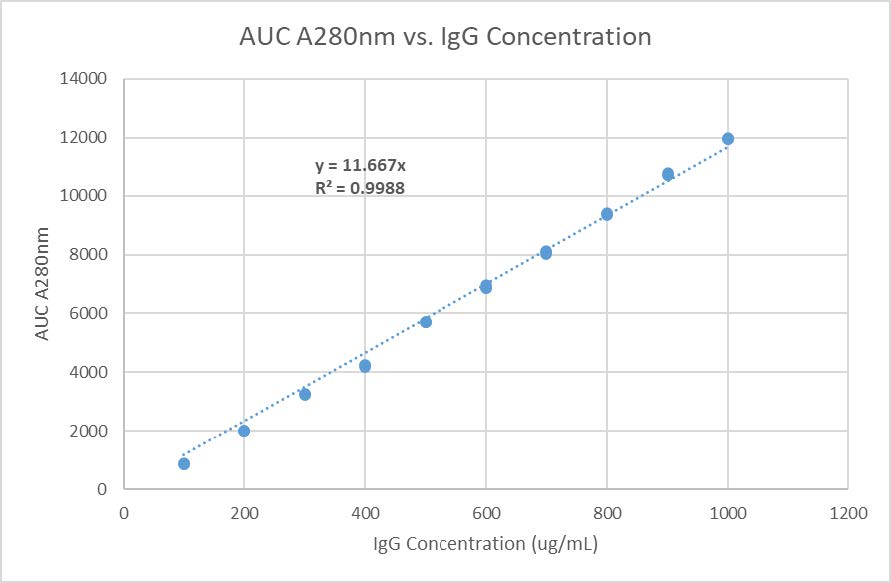
For CROs supporting upstream process development, this allows fast comparison of cell clones, culture media conditions, feeding strategies, and other bioreactor parameters in order to support upstream process development and achieve the highest productivity possible.
3.Clone Screening and Lead Selection
During early development, speed matters more than ever. Protein A HPLC-UV supports high-throughput clone ranking through rapid identifi cation of high producers and parallel assessment of productivity and expression quality. Because the method is largely molecule-agnostic for IgG formats, it avoids the delays associated with assay redevelopment for each new candidate, the biggest source of delays in analytical platforms.
4.Capture Step Evaluation in Process Development
This is the application that sees the most use internally at Rilas!!
Even at the analytical scale, protein A HPLC can model parameters relating to preparative scale protein A runs, including (but not limited to): binding capacity, elution behavior, recovery, and load tolerance.
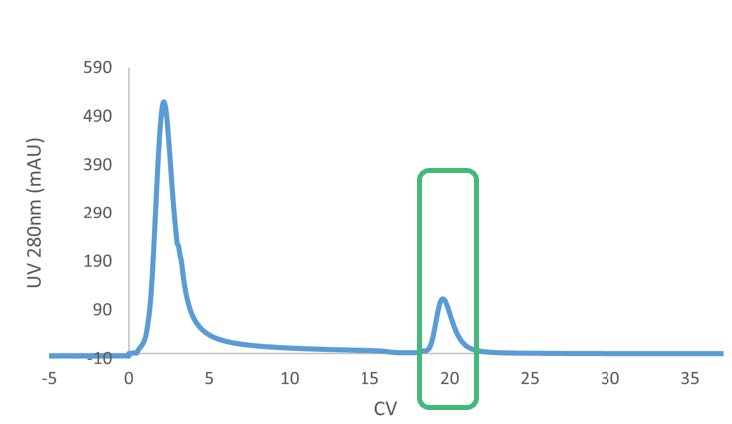
This makes it a valuable tool for process development, including at the steps of: resin screening, buffer optimization, and early-scale-down studies. Data generated analytically often translates well to preparative Protein A steps (moreso than for other forms of chromatography).
5.Comparative Analytics Across Formats
Protein A HPLC-UV is applicable to molecules beyond classical mAbs. While it remains the mainstay of mAb processes, protein A has found utility in the Fc-fusion protein space, the bispecifi c antibody space, the antibody-drug conjugate (ADC) space, and the engineered IgG variant space. Retention behavior, peak shape, and recovery can provide early insight into Fc accessibility and binding characteristics, which may correlate with developability attributes.
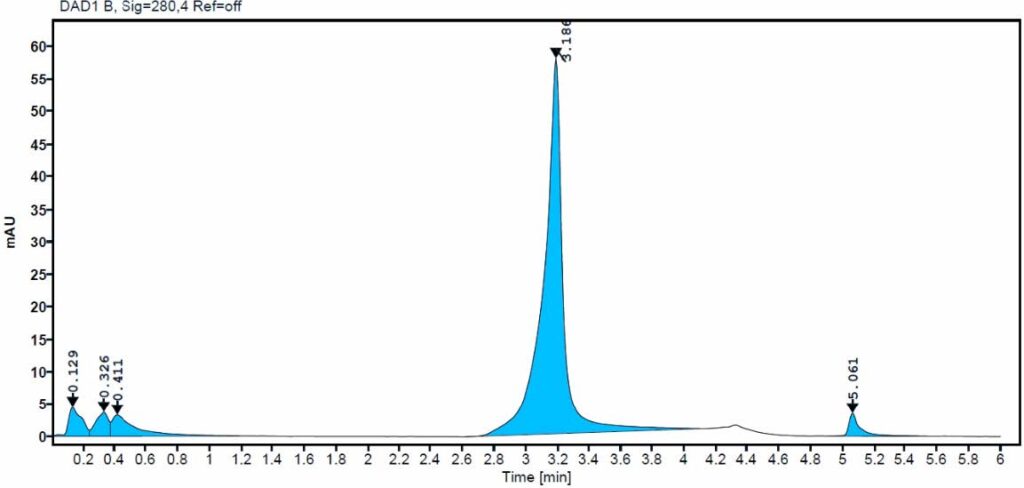
6.Lot-to-Lot and Process Consistency Monitoring
Because the method is highly reproducible, it can be used to compare batches, monitor process changes, and track productivity trends over time. For clients transitioning from discovery into process development, this continuity is critical.
Beyond analytics, Protein A remains a cornerstone of commercial antibody manufacturing, where it serves as the principle platform for downstream processes, as it remains the gold standard capture step for regulatory-accepted purifi cation strategies. By offering Protein A HPLC-UV analytically, we help our clients align early data with later manufacturing realities, reducing costly surprises during scale-up. This enables them to make faster, data-driven decisions while maintaining confidence.
Why Use a CRO for Protein A HPLC-UV?
From our side, providing this as a service means:
- Platform methods are already validated
- No need for clients or teams to invest in specialized columns or development time ($$$)
- Rapid turnaround on crude, real-world samples
- Data interpreted by scientists who understand antibody workflows end-to-end
For our clients, it means actionable antibody data without friction or the need for redevelopment.
Final Thoughts
Protein A HPLC-UV is far more than a niche analytical technique; it’s a highly specific, flexible, and scalable tool that bridges discovery, process development, and manufacturing. By leveraging affi nity-based specifi city rather than general physicochemical separation, it delivers reliable results where other methods struggle, especially in crude samples and early-stage workfl ows. As a CRO, we see it not just as an assay, but as an enabler of faster biologics development and one we’re proud to offer as a core service to our clients!
